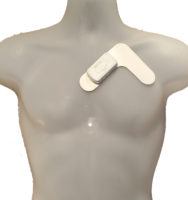
Learn more about the DR400.
No batteries and No SD cards, imagine that!
View hook-up recommendations.
FAQs and links to documents and PCPatch utility.
Innovation
3-Year Warranty
Expertise
Flexible Licensing Models
Thought Leadership
Who We Are
Our mission is to develop and manufacture high quality, cost-effective, and innovative solutions for cardiology, respiratory, and pulmonary applications. As the inventor of the world’s first 14-day dual Holter and event recorder, NorthEast Monitoring is committed to producing innovative solutions to help physicians navigate the complex world of ambulatory cardiac monitoring.
Who We Serve
NorthEast Monitoring is committed to advancing Holter technology with our innovative ambulatory cardiac hardware and software solutions. We created the world’s FIRST 14-DAY Holter and event recorder, provide flexible licensing models for our software, and offer an industry leading 3-YEAR warranty. Our experienced team of industry professionals provide world class support for all of our products and
our President, Mark Hubelbank, Ph.D., is a published engineering physicist.